Millions suffer from Asthma attacks triggered by exercise. Within five minutes of starting exercising, people can get short of breath, start coughing and wheezing, such that lung function significantly drops. But, on a high-salt diet, the attack is even worse; whereas, on a low-salt diet, there’s hardly a significant drop in function at all.
To figure out why researchers had them all cough up sputum from their lungs, and those on the high-salt diet ended up with triple the inflammatory cells, and up to double the concentration of inflammatory mediators. But why? What does salt intake have to do with inflammation? We didn’t know, until now.
“The ‘Western diet,’ high in saturated fat and salt, has long been postulated as one potential…cause for the increasing incidence of autoimmune diseases in developed countries…” The rapidly increasing incidence of autoimmune diseases may be due to an overactivation of immune cells, called helper 17 cells. “The development of…multiple sclerosis, psoriasis, type I diabetes,…Sjögren’s syndrome, asthma, and rheumatoid arthritis” have all been “shown to involve [this] Th17-driven inflammation.” And, one trigger for the activation of these Th17 cells may be elevated levels of salt in our bloodstream. “The sodium content of processed foods and ‘fast food’…can be more than 100 times higher in comparison to similar homemade meals.”
And, sodium chloride, salt, appears to drive autoimmune disease by the induction of these disease-causing Th17 cells. A turn out there’s a salt-sensing enzyme, responsible for triggering the formation of these Th17 cells.
Organ damage caused by high-salt diets may also activate another type of inflammatory immune cell. A high-salt diet can overwork the kidneys, starving them of oxygen, triggering inflammation. The more salt they gave people, the more activation of inflammatory monocyte cells, associated with “high-salt intake induced” kidney oxygen deficiency. But, this study only lasted two weeks. What about the long term?
One of the difficulties in doing sodium experiments is that it’s hard to get free-living folks to maintain a specific salt intake. You can do what is called metabolic ward studies, where you essentially lock people in a hospital ward for a few days and control their food intake. But, you can’t do that long term—unless you can lock people in a space capsule. Mars520 was a 520-day space flight simulation to see how people might do on the way to Mars and back.
What they found was that those on a “high-salt diet displayed a markedly higher number of monocytes,” which are a type of immune cell you often see increased in settings of chronic inflammation and autoimmune disorders.
This may “reveal one of the consequences of excess salt consumption in our everyday lives,” since that so-called high-salt intake may actually just be the average-salt intake. Furthermore, there was an increase in the levels of pro-inflammatory mediators, and a decrease in the level of anti-inflammatory mediators—suggesting that a high-salt diet has the “potential to bring about [an] excessive immune response,” which may damage the immune balance, and result in “either difficulty [in] getting rid of inflammation or even an increased risk of autoimmune disease.”
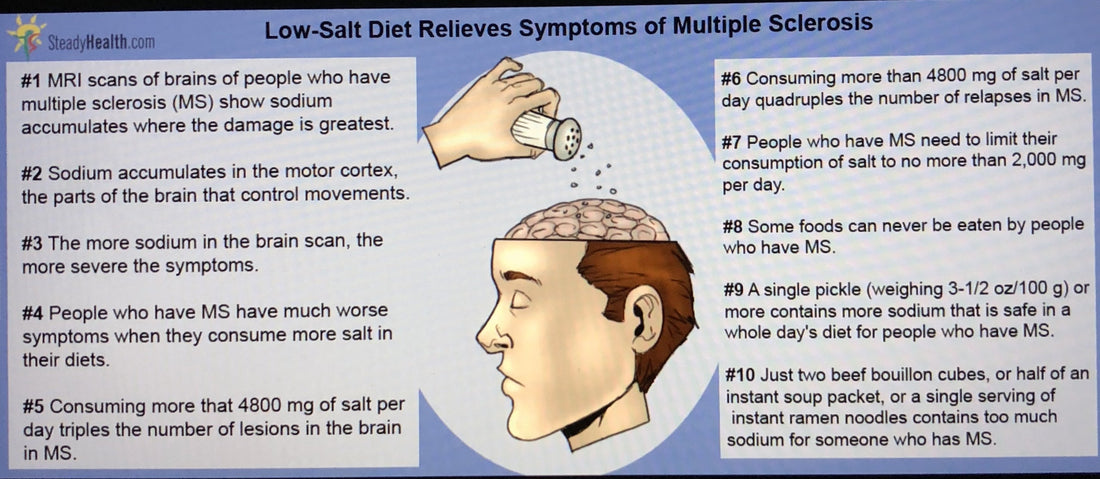
What if you already have an autoimmune disease? “Sodium intake is associated with increased disease activity in multiple sclerosis.” If you follow MS patients for a few years, those eating more salt had three to four times the “exacerbation rate,” three times more likely to develop new MS lesions in their brains, and on average, had eight more brain lesions, fourteen in their brain, compared to six in the low-salt group. So, the next step is to try treating patients with salt reduction and see if they get better. But, since reducing our salt intake is a healthy thing to do anyway, I don’t see why anyone should have to wait. We’re talking especially about White salt.
Team-
Looking Vibrant